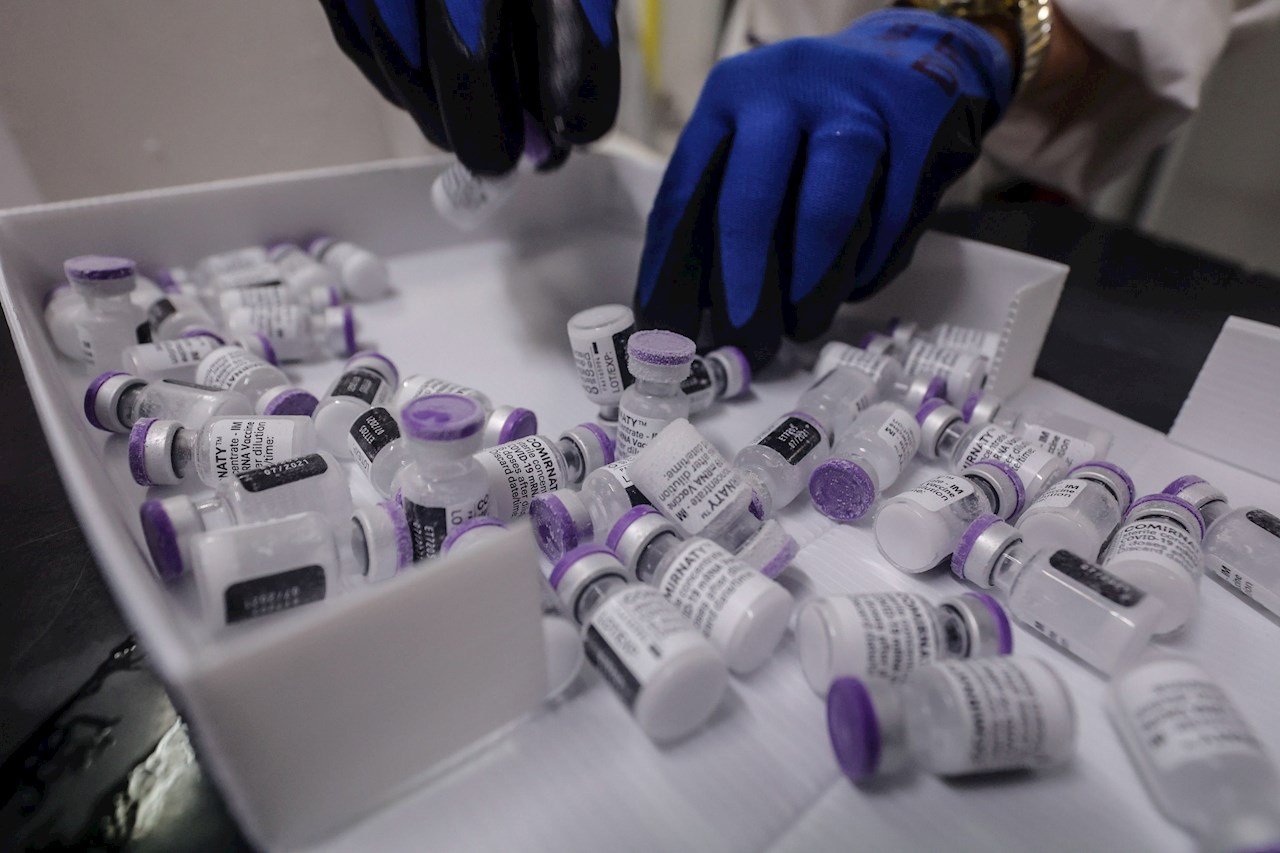
Reuters – Canada and Mexico have imported millions of doses of AstraZeneca Covit-19 vaccine and delivered them to their populations without being properly inspected by the health authorities of the manufacturer’s operations, according to a review of records and regulatory intervention.
The Baltimore plant, which is owned by Emergency Bio Solutions Inc., has been producing Covid vaccines from both AstroGeneca PLC and Johnson & Johnson.
In late March, under pressure to help other countries access the vaccine, President Joe Biden’s administration released the 1.5 million dosage Astrogenogen formula for Canada and the 2.5 million dosage to Mexico.
Officials in both countries told Reuters that European regulators had certified that the Emergency plant complied with “good production practices”, based on which both Canada and Mexico began to use the vaccine.
But the European Pharmaceutical Institute (EMA) told Reuters that the certification was based on a distant study that focused on a part of the facility that did not actually produce AstraZeneca injectables, which had not been announced before.
The U.S. Food and Drug Administration (FDA) stopped production at the factory three weeks later after the J&J vaccine was found to be contaminated with injectable substances.
Follow the information about the world in our international section
Following that, FDA researchers documented unhygienic conditions and poorly trained workers in a factory that was quickly screened to produce vaccines during infections. Millions of doses of both vaccines have been discontinued in the regulatory limbo.
No information about the disease was linked to the vaccines produced by Emergency, and the regulators did not allege that the contaminated vaccines were given to anyone. Emergent said there was no evidence of contamination in the levels of astrogen gene produced on its site.
But the details of the flawed approval process show blind spots that could develop when national regulators share the responsibility of overseeing a complex global pharmaceutical industry. These have been exacerbated by the urgency of the epidemic.
“This is a risk, because who knows the standards in practice?” Said Joel Lex, a Canadian professor and drug regulator. Health Canada generally relies heavily on foreign regulators, he said. “It’s a gap outside of crisis.”
Emergent said in a statement, “We work with health officials around the world to ensure that any vaccine we manufacture meets strict safety and quality requirements before delivery.”
AstraZeneca said Emergency-produced vaccines have been subjected to more than 40 tests to meet its requirements for safety, hygiene and quality, and their production standards are “rigorously evaluated and independently verified by regulators.”
Health Canada told Reuters that it believed the vaccines it had received were safe, in part because of AstraZeneca’s quality control methods. Coferis, Mexico’s health regulator, said he was “confident that the accreditation process was carried out with all the necessary rigors”.
Follow information on business and current affairs at Forbes Mexico
European Inspection
At the beginning of the vaccine distribution, the European Union (EU) was to obtain emergency J&J standards. In February, Italian officials conducted a remote inspection of the plant, which focused only on areas where J&J was produced, “not on the Astrogenega vaccine, but on the EU market,” the EMA said in a statement.
Health Canada said it expects both products to be made to the same standard. “The EMA rated quality systems for this product apply to all similar products manufactured on this site,” it said in a statement.
According to Amir Attar, a professor at the University of Ottawa, this is the wrong approach, including trusting Health Canada’s foreign regulators.
“Cross-contamination risk because they are exactly the same (products),” he said. He said Canada was “entering” by recognizing the astrogenic level of the facility with the FDA.
Health Canada countered that it had tested test results and quality control measures for each batch of vaccines shipped to Canada.
Initially, Coffees of Mexico assured Reuters that the FDA was responsible for certifying US operations, but later confirmed that the factory was certified by European regulators.
When Reuters shared a report from European regulators that they did not particularly evaluate the production of AstraZeneca, Cofribs said it was reconsidering the file, but was optimistic about the approval process.
The Mexican regulator assured that the vaccines went through quality control systems at Emergency and doubled during the bottle filling process handled by another contract manufacturer.
“When evaluating vaccines, the product and its entire production process are considered and tested,” Coffebris said in a statement.
The Emergency Agreement with the U.S. government was the focus of a congressional hearing that uncovered documents revealing early concerns about the Baltimore plant.
Shortly after former President Donald Trump’s administration announced the Emergency Agreement last summer, his own vaccinating officers went to the plant and wrote a report that the staff and training were inadequate. The report warned of the need to strengthen quality oversight, saying the process would “require significant resources and commitment”.
The FDA, after visiting the plant in February 2021, noted in the June note that the revenue of emergency workers was high and that registration was not sufficient. Although European regulators said they were aware of the arrival, Health Canada said it could not find out until last month.
An FDA spokesman said that after inspecting the emergency facilities in April 2021, the company was working closely with international partners and notifying various health officials of its findings.
Follow us on Google News to keep you informed
Emergency said it was working with the FDA to release more volumes of the already-prepared COVID-19 vaccine.
“We have produced tens of thousands of doses that will help people around the world get vaccinated because there are no signs of the disease declining,” the company said.

“Devoted music specialist. Student. Zombie trailblazer. Internetaholic. Food geek.”










More Stories
In Search of Adrenaline: What Kinds of Extreme Tourism to Try
What to Do if Your Laptop Is Warm: 7 Useful Tips
Travel Essentials for a Road Trip